Supercoiling Assays
DNA gyrase is a unique topoisomerase as it is the only enzyme that can catalyse the underwinding of DNA (negative supercoiling). The process is ATP-dependent and occurs through the cleavage and religation of the double-stranded DNA in steps of 2.
For an overview of the reaction please see: , , & (2021). DNA topoisomerases: Advances in understanding of cellular roles and multi-protein complexes via structure-function analysis. BioEssays, 43, e2000286.
A typical reaction is as follows:
1 U of DNA gyrase is incubated with 0.5 µg of relaxed pBR322 DNA in a 30 µl reaction at 37°C for 30 minutes in 1X Assay Buffer
Each reaction is stopped by the addition of 30 µl chloroform/iso-amyl alcohol (24/1) and 30 µl Stop Buffer (STEB: 40% sucrose, 100 mM Tris.HCl ( pH 7.5), 100 mM EDTA, 0.5 mg/ml bromophenol blue), before being loaded on an agarose gel. (0.8%-1.0%: w/v)
Gels can be run either in TBE (90 mM Tris.Borate, 2 mM EDTA) or TAE (40 mM Tris.Acetate, 2 mM EDTA) although the resolution of supercoiled pBR322 is much better if run in TAE at a maximum of 90V for 2 - 3 hours.
Typical Gel:
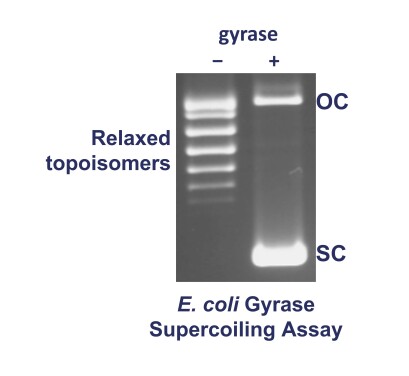
The DNA substrate used (in this case relaxed pBR322) usually consists of a series of bands on an agarose gel. These are topoisomers (DNAs of different linking number).
The treatment of relaxed pBR322 with DNA gyrase converts the relaxed topoisomers to the supercoiled form of the plasmid which migrates faster on an agarose gel. An upper band may also be visible which is open-circular (nicked) DNA which is present in the relaxed substrate but co-migrates with some of the relaxed topoisomers.
Intercalators
Problems can arise in assays if there are contaminants such as ethidium bromide or Chloroquine in the gel tanks. These chemicals are intercalators and can affect the mobility of the DNA leading to confusing results. If there is an intercalator present in the buffer the relaxed topoisomers have increased mobility and run in roughly the same place as negatively supercoiled plasmid. Depending on the amount of intercalator, the negatively supercoiled DNA may have slightly less mobility than normal.
Nucleases
If there are contaminating nucleases present in the assay (due to contaminated assay buffers or impure fractions from cell expression) there may be a significant increase in nicking leading to an increase in the amount of open-circular DNA and possibly the production of linear DNA. This runs as a single band between the OC and SC.
ATP
A loss of activity with previously active enzyme may be due to deterioration of the ATP in the buffer, although, the 5X Assay Buffer has been developed to minimize this. If loss of activity occurs, the addition of extra ATP to the reaction will overcome the problem.
