Decatenation Assays
Type II DNA topoisomerases catalyse the decatenation of inter-linked DNA molecules. During processes such as cell division, they are responsible for unlinking catenated sister chromatids or resolving precatenated or hemicatenated isoforms of DNA during replication. The reaction is ATP-dependent and occurs through the cleavage and religation of the double-stranded DNA.
For an overview of the reaction please see: , , & (2021). DNA topoisomerases: Advances in understanding of cellular roles and multi-protein complexes via structure-function analysis. BioEssays, 43, e2000286.
A typical reaction is as follows:
1 U of enzyme is incubated with 200 ng of kDNA or another catenated substrate in a 30 µl reaction at 37°C for 30 minutes in 1X Assay Buffer
Each reaction is stopped by the addition of 30 µl chloroform/iso-amyl alcohol (24/1) and 30 µl Stop Buffer (STEB: 40% sucrose, 100 mM Tris.HCl ( pH 7.5), 100 mM EDTA, 0.5 mg/ml bromophenol blue), before being loaded on an agarose gel. (0.8%-1.0%: w/v)
Gels can be run either in TBE (90 mM Tris.Borate, 2 mM EDTA) or TAE (40 mM Tris.Acetate, 2 mM EDTA) although the resolution of supercoiled pBR322 is much better if run in TAE at a maximum of 90V for 2 - 3 hours, with or without intercalators (e.g. EtBr or Chloroquine) present.
Typical Gels:

The substrate for decatenation assays is kDNA which consists of a complex interlinked network of catenated DNA maxi- and mini-circles.
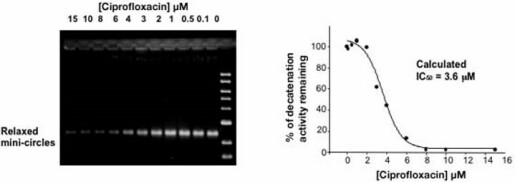
Due to their high molecular mass, these complexes cannot enter an agarose gel under normal electrophoresis conditions, but remain in the wells. In the presence of a type II topoisomerase the mini-circles (2.5 Kb) are released by decatenation and can be resolved quickly and easily at relatively high voltages. Decatenation, rather than relaxation, is the preferred reaction for topo IV and human topo II making this an ideal assay for the quick identification of potential inhibitors.
Intercalators
Intercalators affect the mobility of the DNA during gel electrophoresis. If there is an intercalator present in the buffer the relaxed topoisomers have increased mobility and run in roughly the same place as negatively supercoiled plasmid. Depending on the amount of intercalator, the negatively supercoiled DNA may have slightly less mobility than normal. This is not normally a problem during decatenation, unless you are looking for the relaxed minicircles (as seen in the gel above). But it may in fact be easier to quantify the effect of inhibitors if intercalators are present. As shown in the gel below which is run in the presence of Chloroquine
The IC50 for inhibition of decatenation can be visually assessed as the concentration of compound which leads to a 50% reduction in the mini-circle band. This is then verified using gel documentation software and statistical analysis. In the example below the inhibition of E. coli topo IV- catalysed decatenation of kDNA by ciprofloxacin is followed.
Nucleases
If there are contaminating nucleases present in the assay (due to contaminated assay buffers or impure fractions from cell expression) there may be a significant increase in nicking leading to an increase in the amount of open-circular DNA and possibly the production of linear DNA. This runs as a single band between the OC and SC.
ATP
A loss of activity with previously active enzyme may be due to deterioration of the ATP in the buffer, although, the 5X Assay Buffer has been developed to minimize this. If loss of activity occurs, the addition of extra ATP to the reaction will overcome the problem.
